
Glenorchy
Tasmania 7010 Australia
Phone: 61 3 62497868 (am)
Phone: 61 3 62713000 (pm)
Fax: 61 3 62730010
www.tececo.com
 |
497 Main Road Glenorchy Tasmania 7010 Australia Phone: 61 3 62497868 (am) Phone: 61 3 62713000 (pm) Fax: 61 3 62730010 www.tececo.com |
![]() Printed in cyberspace on recycled electrons
Printed in cyberspace on recycled electrons
Keeping you informed about TecEco sustainability projects. Issue 70, Sept 6th, 2007
Infrastructure Australia 2007 is a highly specialised trade exhibition focusing on the products & services required for commercial, industrial and major projects and John will be talking at 5.00 pm on Friday 7th September 2007 about water capturing Permecocrete pervious concrete which because it uses Eco-Cement has the potential to sequester significant carbon.
Because it is hoped to as much as possible use recycled aggregates to make Permecocrete concrete cannot get much more sustainable.
Used in cities permecocretes will result in significant water capture as well as sequestration. There are many other benefits including a reduction in hot city syndrome and coastal pollution.

John is appearing on behalf of TecEco's Victorian representatives Asset Rehabilitation and Sustainability Services (ARSS) and will also be in attendance at stand D40 (ARSS) from around 1:30 pm on Friday 7th September until 5.00pm.
The proprietors Andy Ward or Leigh Unsworth may be contacted on 95970611. Andy's mobile is 0411088467 and Leigh's mobile is 0408504087.
Whilst in Melbourne John may be contacted on 0413993911.
Unlike conventional additives there are many other advantages of adding reactive magnesia including in particular a marked reduction in water related shrinkage and substantially improved rheology.
There are many in the world investigating the use of Robotics for construction and mixes that are as plastic as a result of the addition of reactive magnesia are very suitable for this purpose as they exhibit what is probably best described as a non Newtonian pseudoplastic fluid properties with viscosity decreasing as shear rate increases in accordance with a Power Law or similar model.
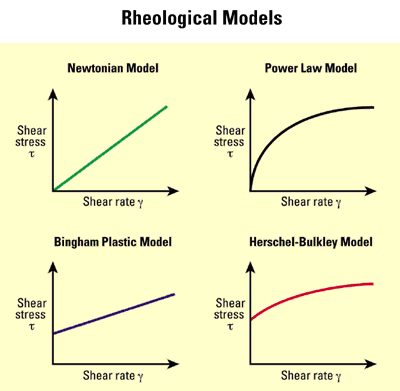
TecEco cements containing reactive magnesia[1] also have low sag and high stick. They do not bleed much, are easy to finish and have low shrinkage. There are chemical and physical reasons for these characteristics.
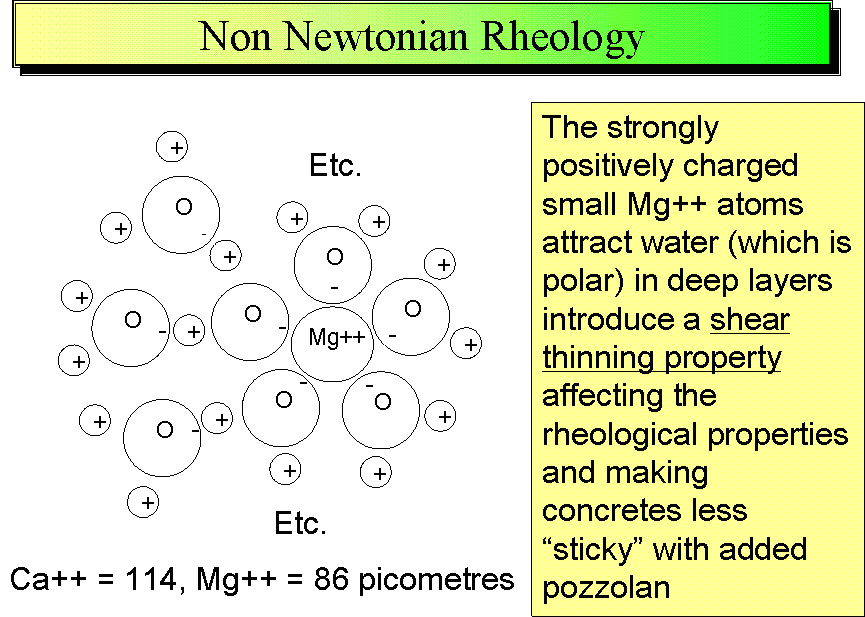
The small highly charged magnesium ion has a strong influence on the rheological and other properties of TecEco magnesium cements including the pseudoplastic thixotropic nature, low bleed, low sag, high stick and low shrinkage. Water, being a polar molecule with a differential charge density and one of the main ingredients of Portland cement concretes when freshly mixed, is strongly affected. Once set strong polar bonding results as the highly charged magnesium ion causes strong differential surface charge densities in the compounds it forms. This tendency for strong polar bonding expresses in the structure of brucite, nesquehonite and other magnesium carbonates and is the reason for their ability to form a wide range of nanocomposites as well as form strong bonds to other surfaces thus increasing the range of waste materials that can be added to concrete composites to advantage.
The viscosity of a liquid is determined by the ease with which molecules can move relative to each other. It depends on the forces holding the molecules together and in water this force is known as hydrogen bonding which is a special case of polar or dipole bonding. Hydrogen bonding describes the attraction between the hydrogen on one polarised water molecule and oxygen on another and is described more fully below because of its importance to rheology and other properties.
Ions in water are either kosmotropic or chaotropic based on their "water structuring" or "water disrupting" nature. Water has cohesivity due to a network of extensive three-dimensional hydrogen bonding and this property is strengthened both by Brucite surfaces and the strongly kosmotropic magnesium ion in solution.
In a recent personal communication Prof Chris Cuff[2] said “Dynamic models of the transient structural effects of magnesium ions in solution, indicate that it and its hydration sheaths are relatively large and also contain extensive arrays of oxygen and hydrogen dipoles and associated networks of hydrogen bonding …..The relatively large size of a transient cluster of water molecules around a magnesium ion (the hydration sheath) imparts a degree of structure not only to itself but also, by conventional electrostatic considerations, to the associated anions in solution. This “structural ordering’ of the solution phase may also lead to drastic effects on the thermal vacancies present in the water. This in turn will lead to major effects on the internal transport processes of the fluid phase. Additionally, due to these constraints, these interactions may lead to the formation of thermodynamically metastable phases many of which may have very strong but metastable internal chemical bonding.”
In relation to the influence of Brucite surfaces on proximate water Wang and others concluded that "The oxygen and hydrogen atomic densities and H2O dipole orientations vary significantly with distance from the surface and deviate from the corresponding structural properties of bulk water to distances as large as 15 A° (~5 molecular water layers) from the surface." (Wang, Kalinichev et al. 2004)
According to Lippmann (Lippmann 1973), and conventional crystallographic electrostatic theory, the ion dipole bond energy, decreases proportionally to the square of the bond length:
E = − e.µ/ r2 .cos(µ, r)
Where e is the ion charge, µ the dipole moment, r the distance between cation center of the negative charge of he dipole (the oxygen center for H2O), and (µ, r) the angle formed by the dipole direction µ and the join cation-oxygen r. The values obtained are 44.9 kcal/mol for Ca2+ and 57.7 kcal/mol for Mg2+. The dipole bond energy of Mg++ is 28.5 % greater than that of calcium and together with the affect of the surface chemistry of brucite is probably sufficient to explain the higher viscosity at low stress (when no "work" is applied as during placement).
According to Wikipedia “A hydrogen bond is a special type of attractive interaction (perhaps a variation of a dipole-dipole bond) that exists between an electronegative atom and a hydrogen atom bonded to another electronegative atom. This type of bond always involves a hydrogen atom, thus the name. Hydrogen bonds can occur between molecules (intermolecularly), or within different parts of a single molecule (intramolecularly).[2] The typical hydrogen bond is stronger than van der Waals forces, but weaker than covalent, ionic and metallic bonds. [3]
In the presence of magnesium ions another type of dipole or polar bond develops between strongly positively charged magnesium ions (Mg++) and the negatively charged oxygen atoms in water molecules which are attracted to them. In solution weakly bonded "hydration shells" many layers deep form around magnesium ions which have a much stronger influence in solution on water than calcium ions and for that matter many dispersants. These sheaths are in addition to the more common primary and secondary hydration shells so important in the kinetics of surface-controlled reactions.
Mg-O dipole bonds are very strong and they distort the electron cloud on the water molecule further towards the oxygen end strongly enhancing hydrogen bonding. The increased strength of hydrogen bonds then propagates (all charges must be statistically equal and opposite) through water and between water and sand or cement as well as additives such as dispersants.
Another way of considering the above is that the strong positive charge density on magnesium ions distorts the electron cloud around water, increasing the density of electrons around an associated oxygen atom in a dipole pair, exposing the hydrogen atoms in water molecules with even less electrons and thus rendering them more positively charged to more strongly hydrogen bond with other oxygen atoms.
Although a relatively large ion in solution due to hydration effects, crystallochemically as Mg++ is a small ion with a diameter, although dependent on site co-ordination and symmetry, of around 86 picometres compared to calcium for example of around 114 picometres. It also has a high bond energy of 57.7 kcal/mol. Compared to other alkali earth metal ions Mg++ has a very high charge density and attaches to oxygen in a similar way to the hydrogen attach to the oxygen of water.
The development of hydration sheaths around magnesium coupled with the interactions with Brucite surfaces with water increase the thixotropic pseudo-plastic property of what amounts to a highly electrostatic fluid that is most importantly very slippery to the bull trowel and float and thus easy to finish.
Although very difficult to determine, the strength of the dipole bonding system described above could be, statistically double that in normal water as a result of the presence of magnesium ions and this strong molecular charge interaction between the magnesium ion and oxygen significantly affects the rheology of the mix.
Still concretes, freshly made with either Tec or Eco-Cements show first set quickly due to the propagation of stronger than normal dipole bonding and interactions with Brucite surfaces with water. When stress (such as during placing) is applied, the electrostatic influence of magnesium ions de-propagates and the weak dipole bonds break up leaving very small balls of water surrounding magnesium ions that are positively charged on the surface and thus repel each other, sand and cement sufficiently to increase fluidity. The better particle packing achieved by using smaller magnesia particles also starts to show influence.
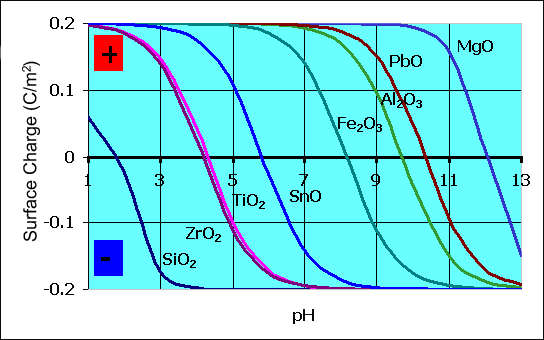
Another important factor that affects early tensile strength gain is that the average surface charge of metal oxides changes with pH and in the case of magnesium oxide goes negative at around pH 12.2 becoming strongly attracted to both sand and cement particles rather than mutually repulsive. Early setting is important because finishers push this requirement in the market place mainly so they can go home earlier.
Not all the affects are electrostatic as the physical size of the magnesium ion added also affects rheology. The mean particle size of the magnesia TecEco generally use is around 8 micron. Experimental work backed up by the mathematical theory of Larrard (de Larrard 1999) indicates that this is not far off the right size for ideal packing with Portland cement which in Australia has a mean particle size of around 20 um[5]. Assuming spheres Larrard calculates a ratio of 1:2.41 (de Larrard 1999).
A further advantage of adding reactive magnesia is a reduction in shrinkage. As concrete sets plastic shrinkage is caused by the losses of water through bleeding followed by evaporation or by seepage if the substrate is permeable (Gani 1997). Neville (Neville 1995)describes drying shrinkage as being the losses of adsorbed water. He stresses that "the loss of free water, which takes place first, causes little or no shrinkage. As drying continues, adsorbed water is removed and the change of volume . . . is in the order of 10000 × 10^6." Both have in common loss of water.
Our original theory to explain the reduced shrinkage of Tec-Cement concretes was that the main cause was the volumetric expansion of magnesium oxide using up free water preventing bleeding by consumption and greater density. Whether this is so and to what extent depends on where the water is coming from during setting and curing and we think that in non permeable concretes containing magnesium it comes from water that would most likely bleed or evaporate out of concrete if it were not for the presence of magnesium.
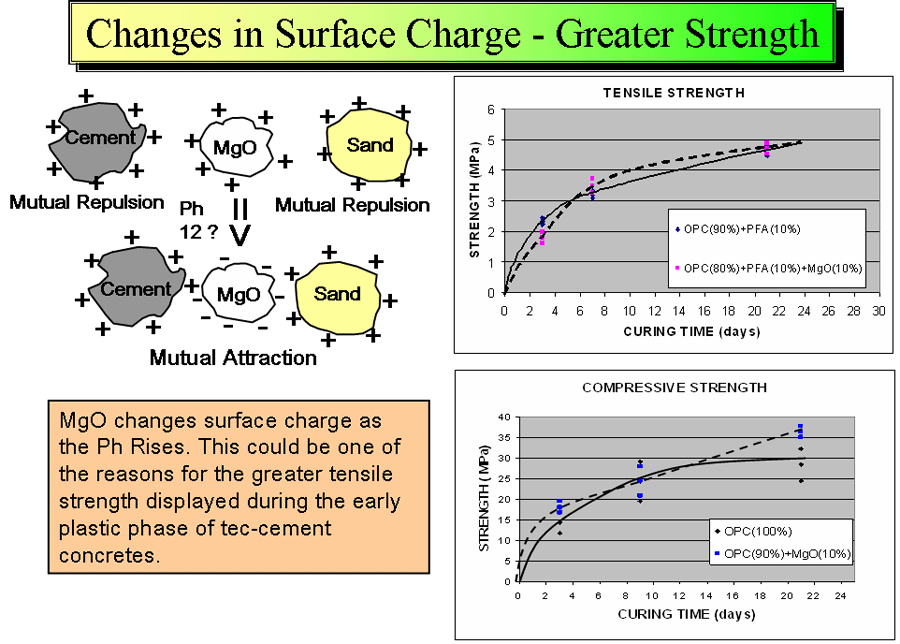
introducing a thixotropic pseudoplastic behaviour property to water reducing its mobility inside concrete (and thus reducing bleeding), the presence of magnesium ions and Brucite have other effects that act together to reduce plastic and drying shrinkage.
Water has an abnormally high surface tension. Water molecules at the liquid-gas surface have lost potential hydrogen bonds directed at the gas phase and are pulled towards the underlying bulk liquid water by the remaining stronger hydrogen bonds. The addition of the strongly charged kosmotropic magnesium ion in sufficient concentration increases this effect and increases surface tension thereby reducing evaporation.
Bleed water does not so readily exit TecEco cement concretes and this must be explained. Water in fresh ordinary Portland cement concretes has a strong capillary action and shortly after it is placed "percolates" through it to the surface where it evaporates or forms bleed water. If concrete is not sufficiently covered or protected by surface coatings of for example aliphatic alcohol, to some extent, depending on the weather it is also pulled by transpiration tension. In such circumstances strong cohesive properties hold the water column in the capillary pores together, preventing tension rupture. The addition of magnesium ions may sufficiently increase the viscosity of water preventing transport through such capillary pores, rendering the capillary water to exposed surface transport system in "normal" concrete, less effective.
Water has an anomalously high enthalpy (44.8 kJ mol -1) and entropy of vaporisation (109 J K-1 mol-1) due to its strong dipole bonding (in water known as hydrogen bonding). The presence of kosmotropic magnesium ions increase the enthalpy and entropy of vaporisation of water, meaning that less would evaporate at the surface again reducing the capillary suction and volume loss. i.e the polar bonds between water and the less volatile magnesium atoms prevents loss of water from the setting mix.
As less water exits concrete containing magnesium ions and Brucite in the above manner the internal relative humidity is maintained at a higher level also reducing later autogenous shrinkage. Longer term drying and autogenous shrinkage may also be reduced because of the surface tightening affect as Brucite carbonates further reduce the loss of internal water and because water is "stored" in brucite hydrates (Mg(OH)2.nH2O species) and available for later hydration.
de Larrard, F. (1999). Concrete Mixture Proportioning: A Scientific Approach, E & FN Spon.
Gani, M. S. J. (1997). Cement and Concrete, Chapman Hall.
Lippmann, F. (1973). Sedimentary Carbonate Minerals. Berlin, Heidelberg, New York, Springer-Verlag.
Neville, A. M. (1995). Properties of Concrete. England, Pearson Education Limited.
Schlumberger. "Oilfield Glossary, X-Y plots of rheological models." from http://www.glossary.oilfield.slb.com/DisplayImage.cfm?ID=373.
Small, R. J., M. L. Peterson, et al. (2005). "Using a buffered rinse solution to minimize metal contamination after wafer cleaning." MicroMagazine.com Retrieved 15 April 2005, from http://www.micromagazine.com/archive/98/01/small.html.
Wang, J., A. G. Kalinichev, et al. (2004). "Molecular modeling of water structure in nano-pores between brucite (001) surfaces." Geochimica et Cosmochimica Acta 68(16): 3351.
[1]Reactive magnesia is also variously known as caustic calcined magnesia, caustic magnesia or CCM. The temperature of firing has a greater influence on reactivity than grind size as excess energy goes into lattice energy and TecEco recommend low temperature calcination
[2]Professor Chris Cuff PhD has worked as a consultant to TecEco and is currently a private consultant in chemical mineralogy and crystallography. Prior to this he was Dean of Science at James Cook University where his tenured appointment was in Geochemistry and Mineral Chemistry. Prior to his appointment at James Cook he was employed by then Process Technology Division of the United Kingdom Atomic Energy Commission working on the crystallisation of phases from high concentration aqueous solutions. Over the last 30 years Prof. Cuff has served on numerous national and international committees including the Council of the Australian Council of Nuclear Science and Engineering and on Committees of the International Mineralogical Association. He has served as assessor for various research councils including ARC and NSF and has acted as peer reviewer for numerous publications in the refereed professional literature.
[3] http://en.wikipedia.org/wiki/Hydrogen_bond
[4]Graphic modified.
[5]Personal communication, Tony Thomas, Chief Cement Engineer, Boral.